Manufacturing and Process OOX deviations are unpredictable. Performing GMP operations without dedicated investigators to prioritize and drive new cases can create delay and waste, since the most efficient approach is immediate root cause analysis to mitigate impact and capture all details.
We provide structured Deviation Investigation services to reduce your costs and organizational frustration, producing ALCOA+ reports by the due date. Our expertise and training ensure investigations are compliant and thorough, with efficient analysis of all root causes, impact, and CAPAs required to prevent recurrence. Our SME investigators excel in recurring cases and complex cases with multiple root causes. And we have experience successfully defending our reports with auditors from the FDA and other regulatory agencies.
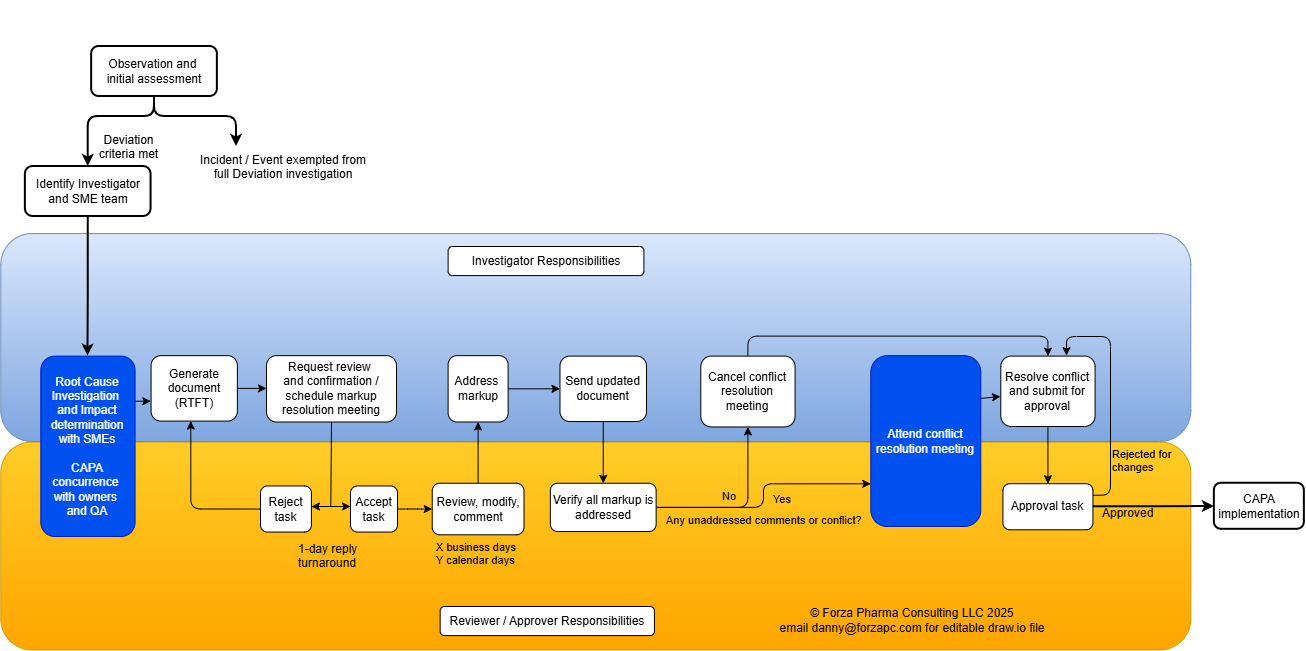
The workflow shown below is typical for approving quality documents. The diagram minimizes the appearance of the investigation itself, as we produce a complete draft that is Right The First Time to minimize review effort. Our network of investigators and library of investigation templates often identifies prior cases with similar causes and relevant CAPAs, even when the event is new to your organization.